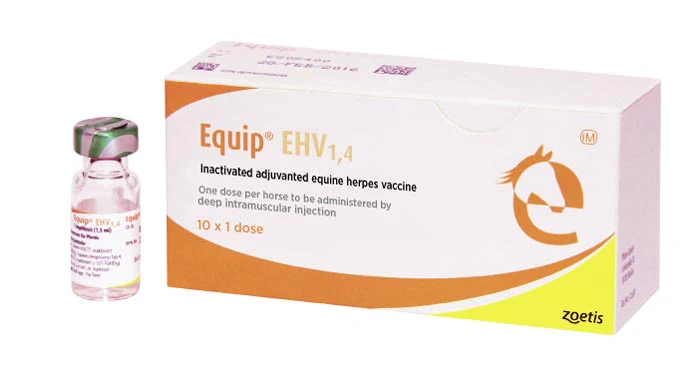
Equip EHV 1,4 Injection
This is a temperature sensitive item, please select the chilled next day delivery option at check out.
For the active immunisation of horses and ponies to reduce the incidence of respiratory disease and viral shedding caused by infection with equine herpes virus types 1 and 4 (EHV-1 and EHV-4) and to reduce the incidence of abortion caused by infection with EHV-1. After completion of the primary vaccination course, the presence of antibodies has been demonstrated within 2 weeks and protection against virulent challenge has been demonstrated within 3 weeks. The duration of immunity is 6 months.
The vaccine has to be stored and transported in its original unopened undamaged packing in the dark at +2°C - +8°C. Exposure to heat and/or direct sunlight has to be avoided. Do not freeze.
Legal category
POM-V
Equip EHV 1,4 Injection
This is a temperature sensitive item, please select the chilled next day delivery option at check out.
For the active immunisation of horses and ponies to reduce the incidence of respiratory disease and viral shedding caused by infection with equine herpes virus types 1 and 4 (EHV-1 and EHV-4) and to reduce the incidence of abortion caused by infection with EHV-1. After completion of the primary vaccination course, the presence of antibodies has been demonstrated within 2 weeks and protection against virulent challenge has been demonstrated within 3 weeks. The duration of immunity is 6 months.
The vaccine has to be stored and transported in its original unopened undamaged packing in the dark at +2°C - +8°C. Exposure to heat and/or direct sunlight has to be avoided. Do not freeze.
Legal category
POM-V
Product Information
Product Information
Shipping & Returns
Shipping & Returns
Description
This is a temperature sensitive item, please select the chilled next day delivery option at check out.
For the active immunisation of horses and ponies to reduce the incidence of respiratory disease and viral shedding caused by infection with equine herpes virus types 1 and 4 (EHV-1 and EHV-4) and to reduce the incidence of abortion caused by infection with EHV-1. After completion of the primary vaccination course, the presence of antibodies has been demonstrated within 2 weeks and protection against virulent challenge has been demonstrated within 3 weeks. The duration of immunity is 6 months.
The vaccine has to be stored and transported in its original unopened undamaged packing in the dark at +2°C - +8°C. Exposure to heat and/or direct sunlight has to be avoided. Do not freeze.
Legal category
POM-V